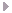| flash spectrum ...photography by Robert B Slobins |
General
Galleries
Details of Second Contact Flash Spectrum, Dundlod - India 24 October 1995

 Previous |
|
 Next |
The flash spectrum lasts just as long in a short eclipse as a long one, if not longer. The arcs are longer and the heavy elements at lower levels are more visible for a longer period of time.
Magnetic fields can lift lighter elements to higher levels than heavier metal elements. The longer curves are of these lighter elements. One can also see the last two bits of the solar photosphere and dark absorption lines crossing their continuous spectra.
The strong red line is Hydrogen-α at 6563 Å. The strong yellow line is Helium I at 5876 Å. Hydrogen-β is the blue line at 4862 Å and the rest of the Hydrogen Balmer series are in the deep blue and violet. Calcium lines are at the far end of the blue and violet at 3970 Å and 3934 Å. In between these major lines one can see shorter lines of elements like Iron, Nickel, Magnesium, Chromium, Vanadium, and Strontium. All of these ions are returning to lower energy levels and emitting light at these discrete frequencies.
The numbers are of wavelengths in Ångstroms (Å): 10-10 meters = 1 Angstrom.